WHAT SHOULD BUSINESSES KNOW ABOUT LEGAL REGULATIONS FOR FOOD SAFETY TESTING?
Food safety testing is not only a mandatory requirement but also a critical safeguard that helps businesses ensure product quality and comply with legal regulations. However, not all organizations fully understand the legal framework, testing standards, or how to select appropriate analytical methods. This article provides a comprehensive overview of legal regulations related to food safety testing, enabling businesses to gain a clear and accurate understanding.
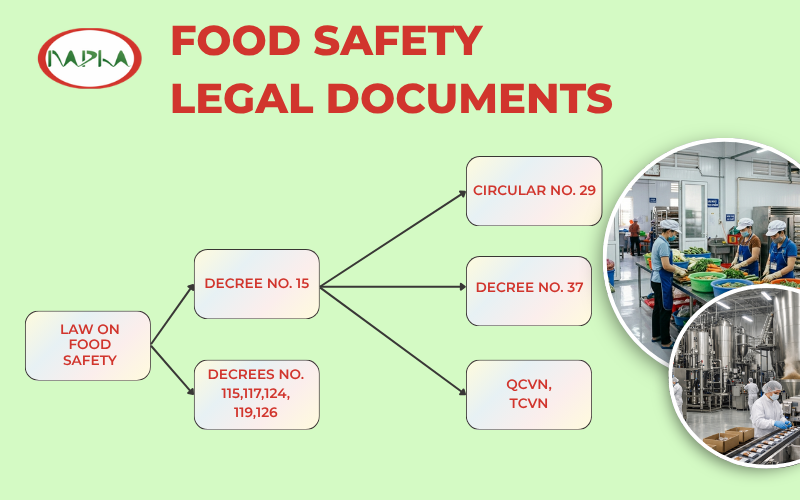
The diagram above provides an overview of the fundamental legal framework governing testing and inspection activities
The Law on Food Safety serves as the primary legal instrument, providing direction for subsequent Decrees (e.g. 15, 115, 117) as well as the current system of Circulars and National Technical Regulations (QCVN/TCVN). It constitutes a mandatory basis for enterprises to benchmark safety criteria and ensure the legal compliance of their products.
II. Food Safety Testing
Food safety testing essentially refers to laboratory analysis activities—a technical process aimed at examining specific parameters of a product.
Nature: The conduct of laboratory testing and analytical procedures to determine quality and safety parameters.
Purpose: To assess whether the product complies with applicable technical regulations or declared standards.
Implementing Authority: Testing must be carried out by laboratories designated by competent state authorities.
1. Basis for Conducting Food Safety Testing
- The quality management system shall comply with TCVN ISO/IEC 17025:2007 or the international standard ISO/IEC 17025:2005;
- Adequate equipment and facilities must be available, aligned with testing requirements and the scope of designation;
- Test methods shall be up to date and properly validated; the laboratory must demonstrate analytical/testing competence for the registered parameters and methods, ensuring compliance with applicable regulations, technical standards, and other relevant requirements as prescribed by the competent line ministry;
1.1. Verification of Accreditation Certificates and Scope of Designation: Not all laboratories accredited to ISO/IEC 17025 are authorised to test all parameters.
- Test Scope: Request a list of accredited parameters. For example, a laboratory may be accredited for microbiological testing but not for heavy metal analysis in your specific food matrix.
- Designation by Competent Authorities: The laboratory must be included in the list of entities designated by the Ministry of Health, the Ministry of Agriculture and Rural Development, or the Ministry of Industry and Trade to support state management of food safety.
1.2. Capability in Handling Complex Sample Matrices
- Example: Testing pesticide residues in vegetables is significantly less complex than in dietary supplements or health protection foods, due to potential matrix interferences.
- Requirement: Confirm whether the laboratory has prior experience testing similar sample matrices. A reputable laboratory should be able to recommend optimal sample extraction methods to minimise analytical errors.
1.3. Basis of Test Methods
- Method Identification: The method column should reference recognised standards such as ISO, TCVN, AOAC, or SMEWW.
- Common Pitfall: Using a test method that is not suitable for the sample matrix. For instance, applying a method designed for bottled water to carbonated beverages may lead to inaccurate results and rejection during regulatory post-inspection.
- Legal Compliance: The applied test method must fall within the scope of methods for which the laboratory has been officially designated by the Ministry of Health or the Ministry of Agriculture and Rural Development.
2. Safety Parameters
Pursuant to Official Dispatch No. 03/ATTP-KN dated 02 January 2019 issued by the Food Safety Authority regarding safety parameters and food safety requirements (e.g. microbiological criteria, heavy metals, mycotoxins), compliance shall be ensured in accordance with the following principles:
1. In accordance with Vietnam National Technical Regulations (QCVNs):
- QCVN 8-1:2011/BYT: National Technical Regulation on mycotoxin limits in food;
- QCVN 8-2:2011/BYT: National Technical Regulation on limits of heavy metal contamination in food;
- QCVN 8-3:2011/BYT: National Technical Regulation on microbiological contamination limits in food;
- Applicable QCVNs for food additives and flavourings;
- Relevant QCVNs for specific food product categories.
2. In accordance with Circulars issued by Ministries and Authorities:
- Circular No. 24/2013/TT-BYT: Regulations on maximum residue limits of veterinary drugs in food;
- Circular No. 50/2016/TT-BYT: Regulations on the list of plant protection products and maximum residue limits of pesticides in food;
- Other relevant circulars and regulatory documents.
3. Local technical regulations applicable to specialty and traditional food products manufactured within specific localities.
4. In the absence of applicable national technical regulations, ministerial circulars, or local technical regulations, the following hierarchy shall apply:
4.1. National Standards (TCVN);
4.2. Standards issued by the Codex Alimentarius Commission, regional standards, or foreign standards;
4.3. Product standards established by the manufacturer, provided that safety parameters are developed based on the aforementioned regulatory framework.
CONTACT FOR FREE CONSULTING VIA HOTLINE: 0938.161.564
NAPHA CONSULTING CO., LTD
Địa chỉ: 3 Floor, An Phu Plaza, 117 - 119 Ly Chinh Thang Street, District 3, HCMC
Email: tuvannapha@gmail.com











main.comment_read_more